|
Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals. The chemical behavior of the various elements is influenced more by the charge of their ion than by any other intrinsic property. For best results, choose Landscape and ‘Fit’ for. The table is available for download in PDF format for offline printing. The position of an element on the periodic table tells about the type of ions it will form, with the elements on the right forming anions (negative charge) and. This periodic table contains the number, symbol, name, atomic mass and most common valence charge of each element. Consequently, beryllium is significantly smaller than lithium. The improved table can be found at Black and White Printable Periodic Table Chart with Oxidation States. This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). acetate PERIODIC TABLE OF IONS arsenate arsenite benzoate borate bromate carbonate chlorate chloride chlorite chromate cyanate cyanide dichromate CH3COO AsO4 3 AsO3 3 C6H5COO BO3 3 BrO3 CO 3 2 ClO3 Cl ClO2 CrO4 2 CNO CN Cr2O7 2 oxalate perchlorate periodate permanganate peroxide phosphate. (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. 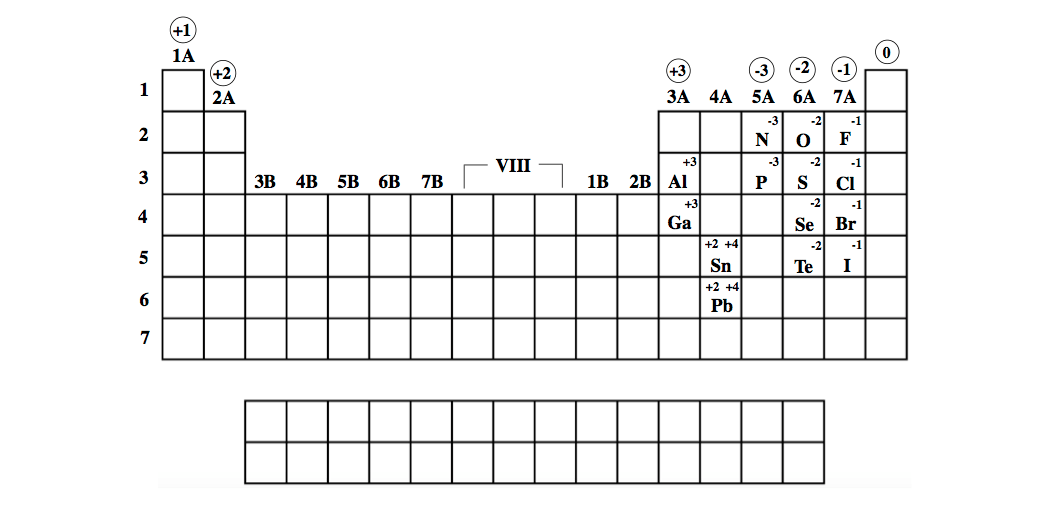 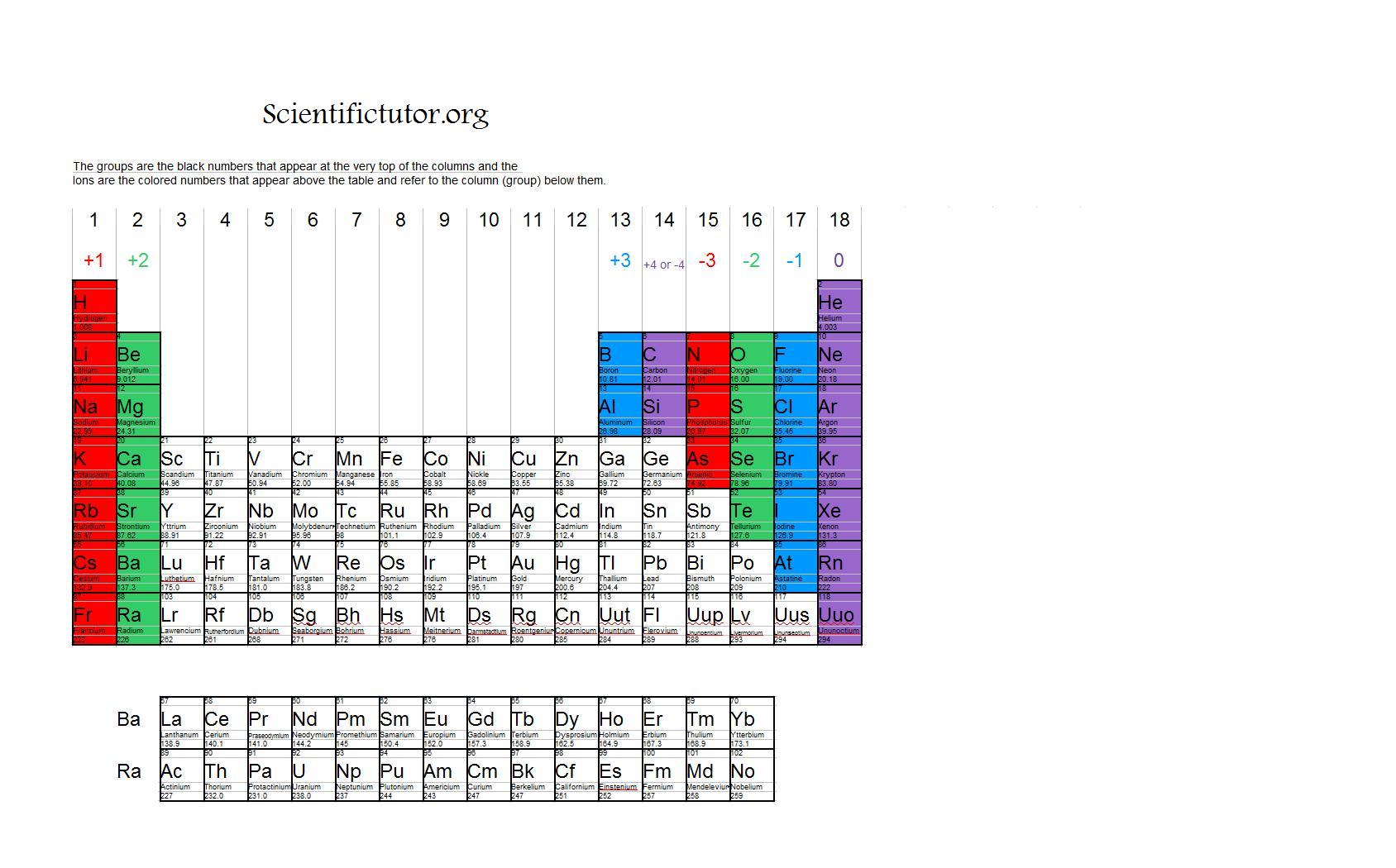
The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. 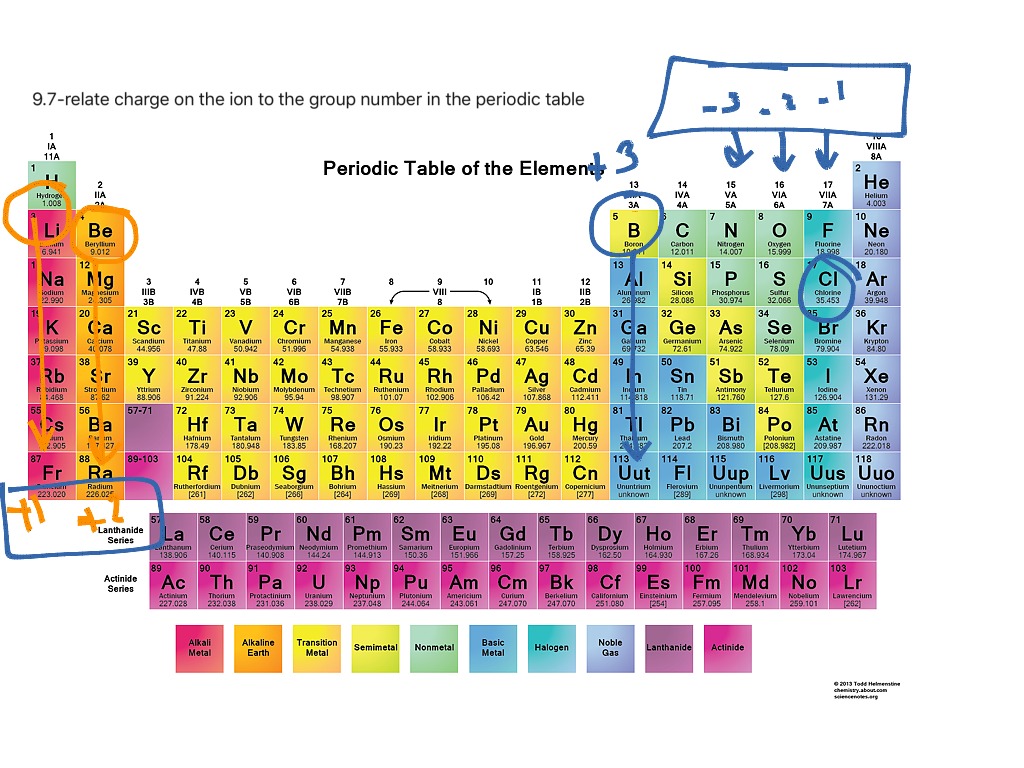
Reviewing the way of finding the charge on an atom may help, too. Figure 3.3 'Predicting Ionic Charges' shows how the charge on many ions can be predicted by the location of an element on the periodic. On the other side of the periodic table, the next-to-last column, the halogens, form ions having a 1 charge. Be sure to memorize silver (Ag + ) and zinc ( Zn 2 + ) charges in addition to this chart.\( \newcommand\)) experienced by electrons in the outermost orbitals of the elements. Ions formed from elements in Group IA of the periodic table have a 1+ charge. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. All Group 17 Elements (halogens) gain one electron to form an ion with a 1. Group 13 metals like aluminum lose three electrons to form an ion with a 3+ charge. 
All Group 2 Elements (alkaline earth metals) lose two electrons to form an ion with a 2+ charge. Within a group (family) of elements, atoms form ions of a certain charge. All Group 1 Elements (alkali metals) lose one electron to form an ion with a 1+ charge. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
